
VectorPro® materials testing software
Designed and developed by Mecmesin specifically for our OmniTest and MultiTest-dV range of materials testers.
Intuitive touch-friendly interfaces and a powerful drag and drop test builder, enabling operators to create everything from complex materials testing procedures to simple product testing, with audit trails and e-signatures supporting FDA 21 CFR Part 11 compliance. Ideal for both production Quality Control (QC) or Research and Development (R&D) environments.

Power and flexibility for your materials testing
Create standards-based tests and your own custom routines
Start measuring stress/strain, tensile, shear, flexure and compressive forces to evaluate your materials. It takes just a few minutes to create your own step-by-step test routines.
We will help get you started and are always on hand to support you.
VectorPro's key features
Design
Intuitive icon-driven interface and workspace
Advanced 'drag and drop' test routine builder
Personalised user accounts and favourites
Touchscreen enabled
Batch testing sets of samples
Control
Stress/strain machine control
Active load control (load holding & load ramping)
System deflection compensation
Manage user permissions and audit trail
Methods for texture analysis
Report
Built-in materials testing calculations
Customisable test report templates
Print and e-mail reports in Adobe PDF format
Export data to Excel and other analysis packages
Support for CFR 21 processes and SQL server database

Precise software control
VectorPro MT testing software runs on Windows 10 computers, taking full control of the test frame motor whilst simultaneously collecting data from the ELS loadcell and displacement channels.
Connect an extensometer to measure elongation directly on the specimen.
Start your chosen routine from a PC or console and VectorPro MT automates the complete test procedure.
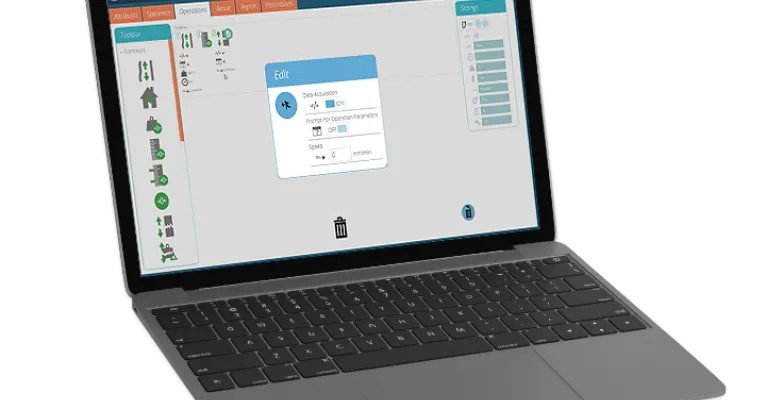
Intuitive test builder
Design and configure step-by-step test routines to match your testing procedures. An intuitive 'drag and drop' interface makes it easy to create, customise and save tests.
VectorPro MT guarantees precision testing by providing stress-strain machine control, basic and active load control, as well as system deflection compensation.
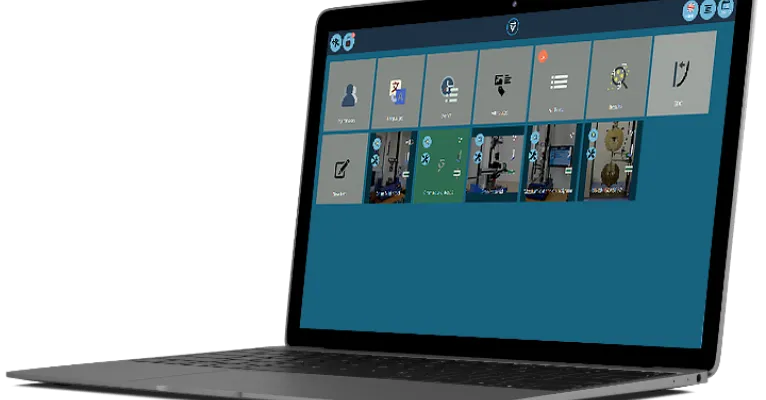
Controls that mean business
Personalised user accounts enable quick access to commonly performed tests, and restricts who can create or run tests, view results and produce reports.
VectorPro MT's event log enables supervisors to authorise actions with e-signatures, providing an audit trail for FDA 21 CFR Part 11 compliance.
Capture essential information with a 'prompt for value' (mandatory user input) feature, before performing a test.
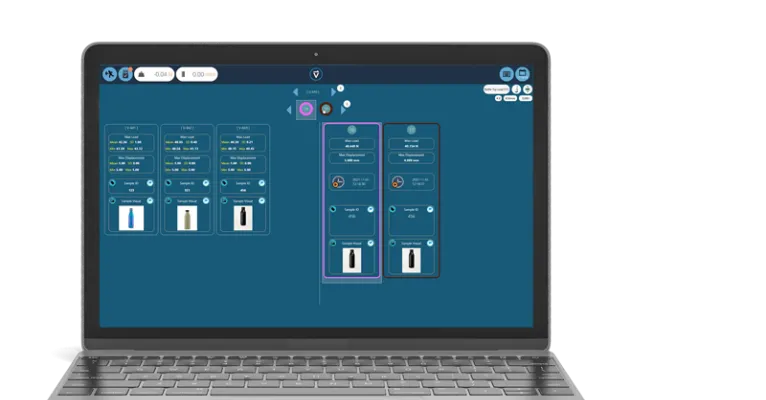
Built for batch testing and automation
Batch testing in VectorPro uses a pre-configured test to create a semi-automatic or fully automatic testing system on sets of products and batches of the products.
It simplifies the use of VectorPro by automatically opening the test and creating a new test with a graph for every sample, exporting data and printing test reports. Test reports can contain overlay graphs or individual test graphs.
This functionality also allows monitoring of the batch progress and can be started, paused and re-started at any time.

Live performance and realtime results
See exactly how your test specimen is performing with realtime results on a live graph, with data captured at 500 times a second (500 Hz).
Easily change graph axes between stress/strain, load/displacement and load/time to better present and understand test results.
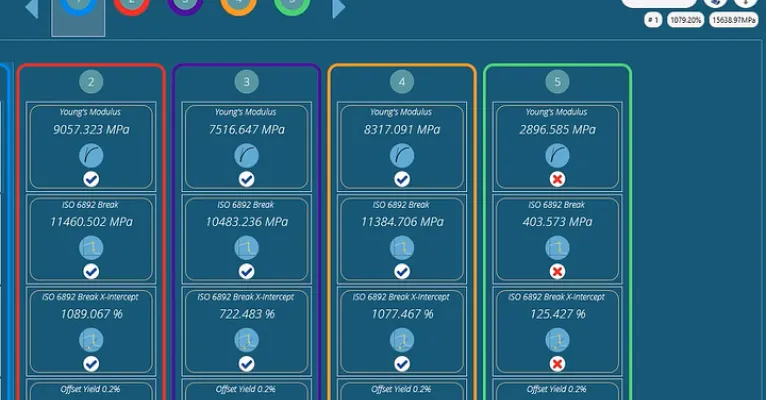
Pass or fail indicator
Specify your own pass and fail criteria within each test, for results that immediately alert operators to the status of each test specimen.
A simple 'pass' (tick) and 'fail' (cross) makes interpreting individual results quick and efficient.

Calcuate results for instant reports and analysis
An advanced and flexible reporting module enables you to configure test reports to present data the way your customer wants to see it.
Built-in calculations for Young's Modulus, Offset Yield, Ultimate Tensile Strength, Nominal Strain and more, makes analysing the data simple.
Customise test report templates with your branding, for your business or your customers.
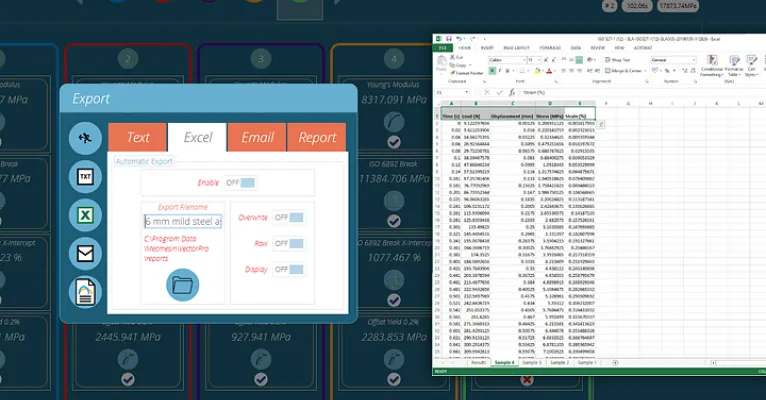
Share and analyse data
All test data can be automatically exported in a format of your choice, from text files and tabulated formats, to Microsoft Excel and email. Data can be exported directly through RS-232 for data-analysis packages.
You can also install VectorPro MT on other computers, for sharing customised test routines and datasets with other users in your organisation.

We're here to help you
Our team of technical engineers, along with our global network of expert partners, will help you get started and show you how to configure routines to your exact testing requirements.
We are always on hand to support you if you need any advice or guidance before, during, or after installation.



























